Organization of JKMA
Organization of JKMA
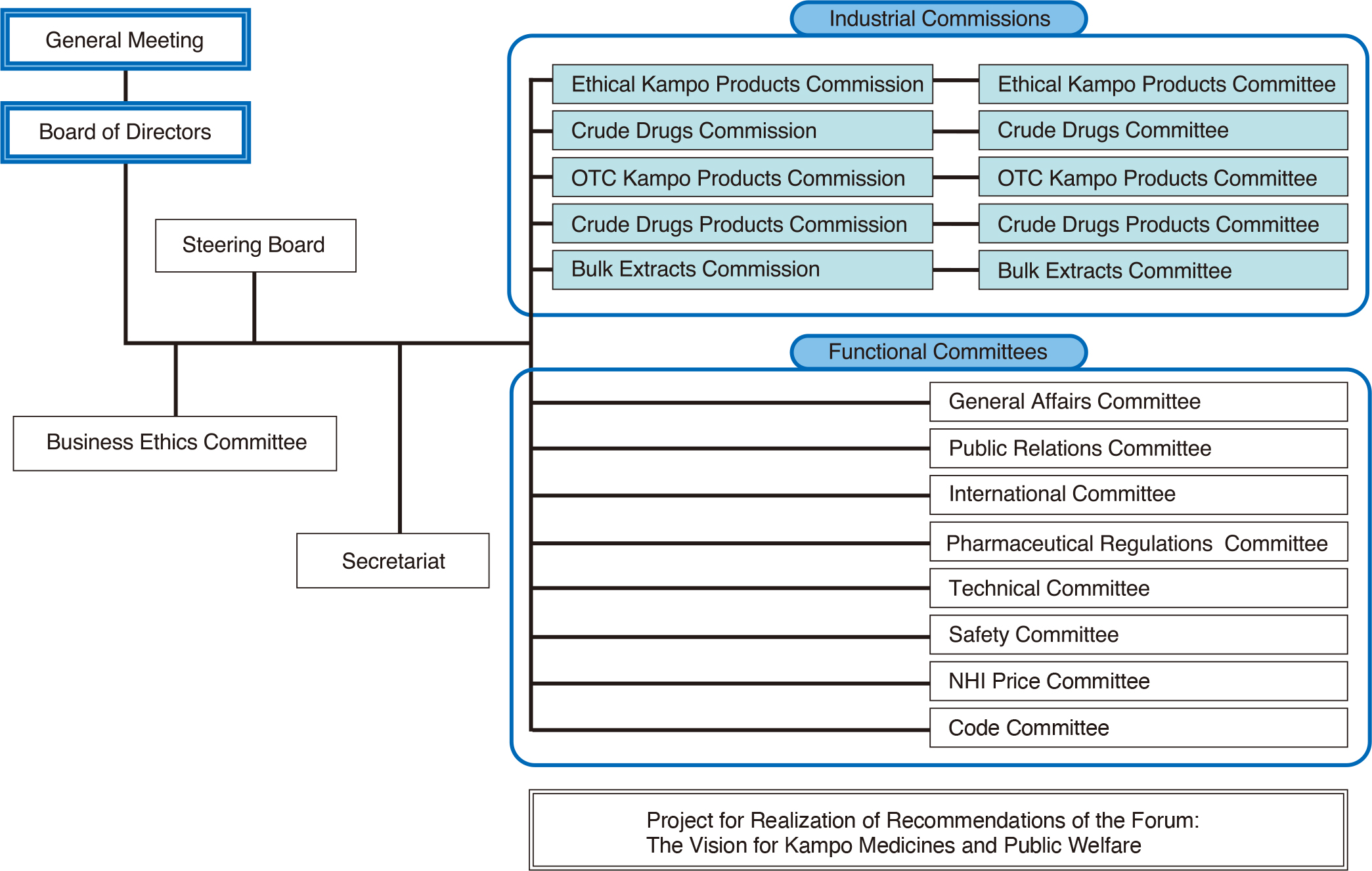
Roles of industrial commissions, projects, and functional committees
■Common matters
- Matters relating to corporate ethics
- Matters relating to cooperation with, negotiations with, and information collection from administrative bodies and related organizations
- Matters relating to business plans and business reports
- Matters relating to budget
Ethical Kampo Products Commission consists of 11 member companies handling ethical Kampo formulation and its bulk extracts.

|
●Chairman Terukazu Kato, Tsumura & Co. |

|
Ethical Kampo Products Committee ●Chairman Hisashi Hasegawa, Tsumura & Co. |
- Nurturing of Kampo products for ethical use and crude drug products
- Matters relating to adherence to fair competition rules and disclosure of Transparency Guidelines
- Matters relating to improvement of distribution
- Matters relating to general training of medical representatives (MRs)
- Matters relating to usefulness of Kampo products for ethical use, such as accumulation of evidence
Crude Drugs Commission consists of 28 member companies handling raw materials for crude drug (captive consumption, sales), and companies handling crude drug for dispensing and other final products.

|
●Chairman Daisuke Tochimoto, Tochimoto Tenkaido Co., Ltd. |

|
Crude Drugs Committee ●Chairman Yutaka Yamamoto, Tochimoto Tenkaido Co., Ltd. |
- Matters relating to quality of crude drugs
- Matters relating to distribution of crude drugs, such as survey on actual distribution of crude drugs for Kampo preparations
- Matters relating to cultivation of crude drugs inside and outside Japan
- Matters relating to revision of Japanese Pharmacopoeia and non-JP crude drug standards in connection with crude drugs
- Matters relating to safety of crude drugs in China, such as survey on use of pesticides
- Matters relating to CITES
OTC Kampo Products Commission consists of 34 member companies handling OTC Kampo formulation.

|
●Chairman Tetsuya Kusayanagi, Kracie Pharmaceutical, Ltd. |

|
OTC Kampo Products Committee ●Chairman Ryuji Takahashi, Kracie Pharmaceutical, Ltd. |
- Matters relating to provision of information on OTC Kampo products to consumers
- Matters relating to standards for approving OCT Kampo product formulations
- Matters relating to training activities for OTC Kampo products
- Matters relating to promotion of proper use of OTC Kampo products
- Matters relating to cooperation on information provision and survey/research on usefulness of OTC Kampo products
Crude Drug Products Commission consists of 24 member companies handling crude drug formulation.

|
●Chairman Kazumasa Kobayashi, Kobayashi Pharmaceutical Co., Ltd. |

|
Crude Drug Products Committee ●Chairman Atsunori Wada, Kobayashi Pharmaceutical Co., Ltd. |
- Matters relating to promotion of development and quality assurance of crude drug products
- Matters relating to information provision and promotion of proper use of crude drug products
- Matters relating to cooperation with the groups and organizations associated with crude drug products
Bulk Extracts Commission consists of 7 member companies manufacturing all types of extracts such as tinctures, dry extracts and so on.

|
●Chairman Shoichi Kuwano, Nippon Funmatsu Yakuhin Co., Ltd. |

|
Bulk Extracts Commission ●Chairman Hiroshi Sasaki, Nippon Funmatsu Yakuhin Co., Ltd. |
- Matters relating to inclusion of Kampo formulations and extracts in official compendiums
- Matters concerning the solvent for manufacturing the crude drug extract
- Matters relating to quality of crude drug extracts
- Matters relating to drug affairs, laws, and regulations concerning crude drug extracts

|
●Project Leader Kenji Ajioka, Tsumura & Co. |
- Matters relating to cross-cutting)/sustainable development of industrial commissions and functional committees in connection with Kampo products, crude drug products, and crude drugs
- Matters relating to realization of recommendations by Forum: The Vision for Kampo Medicine and Public Welfare

|
●Chairman Satoshi Nagano, Tsumura & Co. |
- Matters relating to business policies, business plans, and business reports
- Matters relating to management of budget, account settlement and others
- Matters relating to efficient operation of JKMA activities
- Matters relating to promotion of admission of new members
- Matters relating to compliance
- Matters relating to lecture meetings
- Matters relating to production of Kampo products and others based on crude drugs
- Matters relating to environmental practice
- Cooperation with the related organizations

|
●Chairman Makoto Kitamura, Tsumura & Co. |
- Matters relating to PR for JKMA activities
- Matters relating to educational activities concerning Kampo products and crude drugs
- Matters relating to handling of inquires and interviews by news organizations and the like
- Matters relating to handling of inquires by general consumers, medical professionals and governments
- Matters relating to maintenance and administration of websites

|
●Chairman Hirokazu Koyanagi, Tsumura & Co. |
- Matters relating to international exchange concerning Kampo products and crude drugs
- Matters relating to discussion on responses to changes in international situations and sharing of discussion results in connection with Kampo products and crude drugs
- Matters relating to cooperation in the organizations associated with international issues concerning Kampo products and crude drugs

|
●Chairman Hirokazu Kurita, Kracie Pharmaceutical, Ltd. |
- Matters relating to pharmaceutical affairs systems
- Matters relating to surveys and research on the laws, regulations, and notifications associated with Kampo products and crude drug products

|
●Chairman Yasuhiro Takasugi, Tsumura & Co. |
- Matters relating to quality of pharmaceutical products and raw materials
- Matters relating to listing on Japanese Pharmacopoeia and non-JP crude drug standards
- Matters relating to impurities
- Matters relating to GQP/GMP for pharmaceutical products
- Matters relating to quality in international harmonization (PIC/S, FHH, ICH, and others)

|
●Chairman Masanori Katori, Tsumura & Co. |
- Prompt and thorough execution of safety measures such as revisions of the precautions standardized within JKMA
- Securing of safety and promotion of proper use of Kampo products and crude drug products
- Sharing and discussion of a variety of safety information (general situations and academic information)
- Mutual training among members (lecture meetings and study meetings)
- Cooperation and submission of opinions with administrative bodies and other organizations in the pharmaceutical industry
- Responses to notifications on new points on statements in package inserts for ethical drugs
- Appropriate responses to messages concerning reevaluation results of Kampo products for ethical use
- Discussion on responses to consultation and complaints concerning Kampo products and others based on crude drugs
Code Review Board consists of 5 member companies participating in the Ethical Kampo Products Commission and the Crude Drugs Commission, in order to implement the Promotion Code for ethical Kampo formulation and crude drug.

|
●Representative Member Hiroyuki Matsuzuka, Kracie Pharmaceutical, Ltd. |
- Matters relating to codes
(1)Familiarization with Code of Practice
(2)Promotion of improvement in promotional activities under guidelines for sales information provision
- Matters relating to examination of outline of product information and others
(1)Examination of outline of product information and others and preparation of the board of review report
(2)Thoroughly disseminate the points to be noted when creating product information outlines and Revision and thorough dissemination of information to member companies
(3)Reporting of opinions to and exchange of opinions with administrative bodies, etc.
- Matters relating to Standard for Adequate Advertisement of Pharmaceutical Products
The purpose of the NHI Pricing Committee is to contribute to the improvement of national medical care by ensuring the sustainable growth of ethical kampo formulations and crude drugs within systems such as medical insurance and the NHI drug price standard. The NHI Pricing Committee consists of 15 member companies.

|
●Chairman Makoto Sakaue, Tsumura & Co. |
- Matters relating to survey/research, proposals, and countermeasures in connection with medical insurance systems and drug price standard systems
- Matters relating to cooperation with FPMAJ medical insurance and drug price research council and information collection
- Matters relating to holding of lecture meetings, study meetings, and others